Deaths, Blindness Linked to Recalled Eye Drops Contaminated with Antibiotic-Resistant Bacteria
Along with the death of three individuals, eight individuals have gone blind, and four additional individuals have needed to surgically remove an eyeball after using contaminated eye drops, the CDC said
An antibiotic-resistant bacteria found in multiple recalled eye drops have caused the death of three individuals.
The Centers for Disease Control and Prevention provided an update on their investigation on Tuesday after multiple eye drops distributed by EzriCare and Delsam Pharma and manufactured under Global Pharma Healthcare were recalled in February due to their contamination of the rare Pseudomonas aeruginosa bacteria.
According to their findings, 68 individuals in 16 states who used the contaminated eye drops were infected with bacteria. Along with the death of three individuals, eight individuals have gone blind, and four additional individuals have needed the surgical removal of an eyeball.
Related:Women Who Follow a Mediterranean Diet Could Lower Their Risk of Heart Disease, Report Says
Pseudomonas aeruginosa, explains the CDC, can cause infections in the blood, lungs (pneumonia), and other parts of the body after surgery.
Individuals who previously used the recalled eye drops should look out for symptoms such as yellow, green, or clear discharge from the eye, eye pain or discomfort, and redness of the eye or eyelid. The CDC advises that individuals who present symptoms should seek medical care immediately.
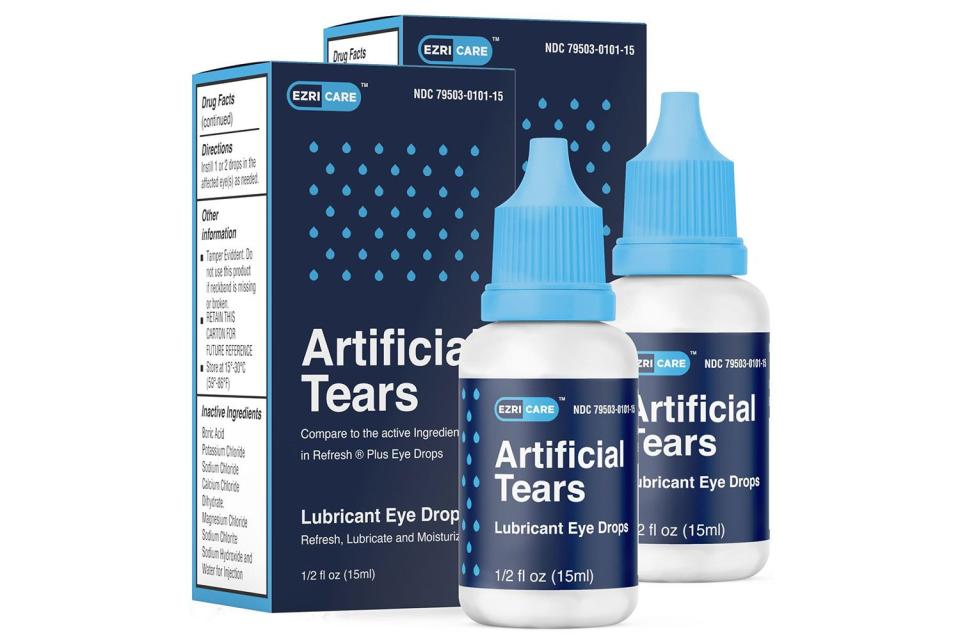
Most cases of Pseudomonas aeruginosa infection were linked back to artificial tear eye drops. The EzriCare Artificial Tears lubricant, a preservative-free, over-the-counter product packaged in multidose bottles, was the brand most commonly reported in infection cases.
RELATED VIDEO: Over 300 Passengers and Crew Members Fall Sick on Ruby Princess Cruise Ship
Never miss a story — sign up for PEOPLE's free daily newsletter to stay up-to-date on the best of what PEOPLE has to offer, from juicy celebrity news to compelling human interest stories.
"Testing of unopened bottles of EzriCare Artificial Tears is ongoing to assist in evaluating for whether contamination may have occurred during manufacturing," shared the CDC in Tuesday's update. "Patients and healthcare providers should immediately stop the use of EzriCare Artificial Tears pending additional information and guidance from CDC and FDA."
While contamination cases are rare, the CDC reports that the bacteria caused an estimated 32,600 infections among hospitalized patients and 2,700 estimated deaths in 2017.
For more People news, make sure to sign up for our newsletter!
Read the original article on People.

 Yahoo Finance
Yahoo Finance 