Deciphera's (DCPH) Q4 Earnings Miss, Qinlock Drives Revenues
Deciphera Pharmaceuticals, Inc. DCPH reported a fourth-quarter 2022 loss of 60 cents per share, wider than the Zacks Consensus Estimate of a loss of 57 cents. In the year-ago quarter, DCPH had reported a loss of $1.51.
Total net revenues were $36.35 million in the quarter, surpassing the Zacks Consensus Estimate of $35.52 million. Revenues rose 50.2% year over year.
Shares of Deciphera have rallied 76.2% in the past year against the industry’s decline of 11.4%.

Image Source: Zacks Investment Research
In May 2020, the FDA approved Deciphera’s sole marketed drug Qinlock (ripretinib) to treat adult patients with advanced gastrointestinal stromal tumors (GIST), who received prior treatment with three or more kinase inhibitors, including Novartis’ Gleevec (imatinib).
Full-Year Results
For 2022, Deciphera generated total revenues of $134 million compared with $96.1 million recorded in 2021, witnessing year-over-year growth of 39.41%.
For the full-year 2022, the company reported a loss of $2.37 per share compared with a net loss of $5.16 per share in 2021.
Quarter in Detail
Apart from Qinlock, there is no marketable drug in Deciphera’s portfolio. Total revenues comprised net product revenues and collaboration revenues. Net product revenues from Qinlock sales were $32.9 million, up 39% year over year. Qinlock generated $25.6 million in sales in the United States and $7.3 million in sales in the ex-U.S. markets.
Collaboration revenues in the reported quarter were $3.5 million compared with $0.5 million recorded in the year-ago quarter.
Research and development expenses were $48 million, down 35.9% year over year owing to lower costs related to clinical studies on Qinlock and the discontinuation of the rebastinib program after corporate restructuring implemented in the fourth quarter of 2021.
Selling, general and administrative expenses were $32.2 million, down 13.3% year over year owing to lower professional and consultant fees.
Deciphera had cash, cash equivalents and investments worth $339 million as of Dec 31, 2022 compared with $327.6 million as of Dec 31, 2021. Deciphera expects its current cash balance together with the anticipated product, royalty, supply revenues and the net proceeds from its underwritten public offering completed in January 2023 to fund its operating and capital expenditures into 2026.
Other Updates
In November 2021, the European Commission approved Qinlock for treating adult patients with advanced GIST, who already received treatment with three or more kinase inhibitors, including Gleevec. The nod in Europe is already generating incremental sales. Qinlock got included in the National Reimbursement Drug List in China for patients with GIST as well as receiving approval in New Zealand in December 2022.
Deciphera expects to initiate the pivotal phase III INSIGHT study to evaluate Qinlock versus sunitinib for the treatment of second-line GIST patients with mutations in KIT exon 11 and 17/18, later in 2023.
Deciphera is evaluating vimseltinib in the phase III MOTION study to treat patients with tenosynovial giant-cell tumors, who are not amenable to surgery. Enrollment in the study is currently ongoing and expected to be completed in the first quarter of 2023 and report results from the study in the fourth quarter of 2023.
This apart, DCPH, in collaboration with Pfizer, Inc. PFE expects to initiate a new study evaluating DCC-3116 in combination with encorafenib and cetuximab in patients with colorectal cancer. Per the terms of the agreement with Pfizer, Deciphera will bear the cost of the study while Pfizer will supply encorafenib free of cost.
Additionally, Deciphera initiated enrollment in three phase Ib dose escalation studies of DCC-3116 in the fourth quarter of 2022, expecting to report data from the study in the second half of 2023.
Deciphera Pharmaceuticals, Inc. Price and Consensus
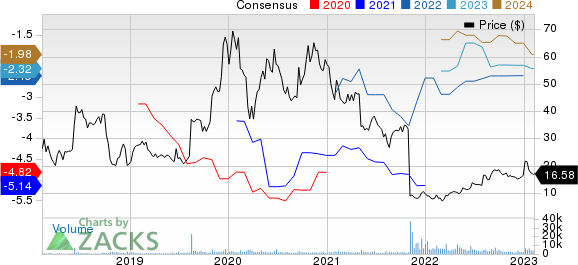
Deciphera Pharmaceuticals, Inc. price-consensus-chart | Deciphera Pharmaceuticals, Inc. Quote
Zacks Rank & Other Stocks to Consider
Deciphera currently carries a Zacks Rank #3 (Hold).
Other stocks worth considering in the biotech sector are 89BIO, Inc. ETNB and ADMA Biologics, Inc. ADMA both carrying a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
In the past 90 days, the estimate for 89BIO’s 2022 loss per share has narrowed from $3.81 to $2.99. During the same period, the loss estimate per share for 2023 has narrowed from $3.59 to $2.59. In the past year, the shares of 89BIO have risen by 170%.
ETNB’s earnings witnessed an average earnings surprise of 10.08%, beating three out of four estimates in the trailing four reported quarters.
In the past 90 days, the consensus estimate for ADMA Biologics’ 2022 loss per share has narrowed from 34 cents to 33 cents. During the same period, the loss estimate per share for 2023 has narrowed from 20 cents to 19 cents. In the past year, the shares of ADMA Biologics have increased by 131.1%.
ADMA’s earnings beat estimates in three of the trailing four quarters, delivering an average earnings surprise of 1.81%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Pfizer Inc. (PFE) : Free Stock Analysis Report
ADMA Biologics Inc (ADMA) : Free Stock Analysis Report
Deciphera Pharmaceuticals, Inc. (DCPH) : Free Stock Analysis Report
89BIO (ETNB) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 