Denali (DNLI) Q2 Earnings Beat, Pipeline Progress in Focus
Denali Therapeutics DNLI incurred a loss of 48 cents per share in second-quarter 2022, narrower than the Zacks Consensus Estimate of a loss of 52 cents and the year-ago quarter’s loss of 50 cents per share.
Collaboration revenues came in at $52.5 million in the reported quarter, beating the Zacks Consensus Estimate of $45 million and up from $22.9 million in the year-ago period. The increase was primarily due to a $12.0 million preclinical milestone earned for approval of the CTA for TAK-920/DNL919 (ATV:TREM2) in the second quarter and $40.0 million of milestone revenues earned in April 2022 upon dosing the first patient in a phase II study of SAR443820/DNL788 in individuals with amyotrophic lateral sclerosis (ALS).
In the year so far, Denali’s shares have lost 16.5% compared with the industry’s 17.3% decline.
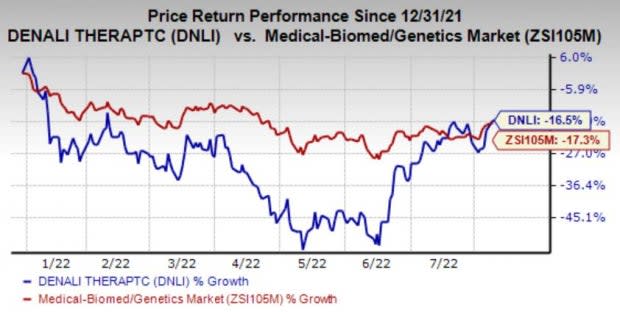
Image Source: Zacks Investment Research
Quarter in Detail
Research and development expenses were $92.7 million in the quarter under review compared with $65.7 million in the year-ago period.
General and administrative expenses were $21.2 million, up from $19 million in the year-ago quarter.
Cash, cash equivalents and marketable securities were approximately $1.16 billion as of Jun 30, 2022.
Denali Therapeutics Inc. Price, Consensus and EPS Surprise
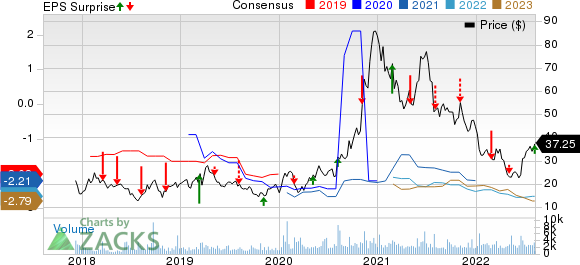
Denali Therapeutics Inc. price-consensus-eps-surprise-chart | Denali Therapeutics Inc. Quote
Recent Updates
Denali has collaborated with Biogen BIIB to advance its LRRK2 inhibitor program to address Parkinson’s disease (PD). In May 2022, Denali and Biogen announced that dosing had commenced in the global phase IIb LUMA study to evaluate the efficacy and safety of BIIB122 compared to placebo in 640 participants with early-stage PD.
The phase III LIGHTHOUSE study in approximately 400 participants with PD with a confirmed LRRK2 pathogenic variant is expected to begin in the second half of the year.
The company has also partnered with Sanofi SNY for its two separate receptor-interacting serine/threonine protein kinase 1 (RIPK1) inhibitor programs to address neurological diseases using the central nervous system (CNS)-penetrant RIPK1 inhibitors and peripheral inflammatory diseases using non-CNS penetrant RIPK1 inhibitors.
In May 2022, Sanofi began dosing with SAR443820 in the phase II HIMALAYA study, which is expected to enroll approximately 260 participants with ALS. Consequently, Denali received a $40 million milestone payment from Sanofi in the second quarter related to the initiation of the phase II HIMALAYA study.
Denali has initiated recruitment of phase II/III COMPASS study, which will enroll approximately 54 MPS II (Hunter syndrome) patients. Upon completion of the ongoing phase I/II study, and together with data from the global COMPASS study, this combined data package will potentially support registration,
Zacks Rank
Denali currently has a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Sanofi (SNY) : Free Stock Analysis Report
Biogen Inc. (BIIB) : Free Stock Analysis Report
Denali Therapeutics Inc. (DNLI) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 