Ionis (IONS) Posts Two-Year Upbeat Data From HAE Drug Study
Shares of Ionis IONS increased 5% on Jun 1, after management announced positive clinical progress with donidalorsen, its late-stage investigational prophylactic therapy for hereditary angioedema (“HAE”).
Management revealed top-line data from an ongoing phase II open label extension (“OLE”) study, which showed that study participants treated with donidalorsen achieved an overall sustained mean reduction in HAE attack rates of 96% from baseline over a two-year period across dosing groups. The company plans to present these results at future medical meetings.
A rare genetic disease, HAE is marked by severe and potentially fatal swelling of the arms, legs, face and throat.
Ionis also announced that it has completed the enrolment in the phase III OASIS-HAE study, which is evaluating the safety and efficacy of donidalorsen in preventing HAE attacks and the drug’s impact on quality of life. The top-line data from this study is expected in first-half 2024.
In the year so far, shares of Ionis have risen 13.7% against the industry’s 4.9% fall.
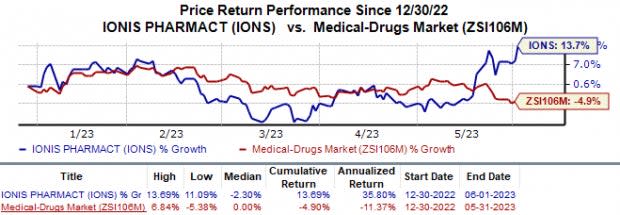
Image Source: Zacks Investment Research
Overall, Ionis has phase III studies ongoing for seven medicines (internal and partnered) across nine indications, including donidalorsen for HAE
Some of the other candidates are tofersen for SOD1-ALS (amyotrophic lateral sclerosis), pelacarsen for cardiovascular disease due to elevated Lp(a) levels, olezarsen for familial chylomicronemia syndrome (FCS) and severe hypertriglyceridemia, eplontersen for TTR amyloidosis, ION363 for ALS, with mutations in the fused in sarcoma gene, or FUS; and bepirovirsen for chronic hepatitis B.
Biogen BIIB, AstraZeneca AZN, Novartis and GSK are its partners for tofersen, eplontersen, pelacarsen and bepirovirsen, respectively. Ionis and AstraZeneca’s new drug application seeking approval of eplontersen for polyneuropathy caused by hereditary TTR amyloidosis (ATTRv-PN) is under review with the FDA with a decision expected on Dec. 22, 2023.
Ionis and AstraZeneca are also developing eplontersen for treating cardiomyopathy in the phase III CARDIO-TTRansform study. AstraZeneca/Ionis expects to complete enrolment in this study in mid-2023.
Last month, the FDA granted accelerated approval to Biogen/Ionis’ tofersen for treating superoxide dismutase 1-amyotrophic lateral sclerosis. The drug will be marketed by Biogen under the trade name of Qalsody. A regulatory application for the Biogen-partnered drug is under review in the EU.
Donidalorsen is one of the company’s wholly-owned pipeline candidate. Some other of Ionis’ wholly-owned pipeline candidates — olezarsen and ION-363 — are being evaluated in late-stage studies. Data from the phase III BALANCE study of olezarsen in FCS is expected in the second half of this year.
Ionis Pharmaceuticals, Inc. Price
Ionis Pharmaceuticals, Inc. price | Ionis Pharmaceuticals, Inc. Quote
Zacks Rank & Stock to Consider
Ionis currently carries a Zacks Rank #3 (Hold). A better-ranked stock in the overall healthcare sector is Ligand Pharmaceuticals LGND, which sports a Zacks Rank #1 (Strong Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.
In the past 30 days, the estimate for Ligand’s 2023 earnings per share has increased from $4.16 to $5.25. During the same period, the earnings estimate per share for 2024 has increased from $4.58 to $4.69. In the year so far, the shares of Ligand have risen 6.6%.
Ligand Pharmaceuticals beat earnings estimates in two of the last four quarters, while missing the mark on the other two occasions. On average, the company’s earnings witnessed an earnings surprise of 21.50%. In the last reported quarter, LGND delivered an earnings surprise of 121.36%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
AstraZeneca PLC (AZN) : Free Stock Analysis Report
Biogen Inc. (BIIB) : Free Stock Analysis Report
Ligand Pharmaceuticals Incorporated (LGND) : Free Stock Analysis Report
Ionis Pharmaceuticals, Inc. (IONS) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 