Novartis (NVS) Set to Advance Malaria Drug Into Phase III
Novartis AG NVS and partner Medicines for Malaria Venture announced a decision to advance ganaplacide/lumefantrine — solid dispersion formulation (SDF) — into late-stage development for the treatment of patients with acute uncomplicated malaria due to Plasmodium falciparum.
Ganaplacide is a novel agent with a new mechanism of action that is combined with a new formulation of lumefantrine optimized for once-daily dosing.
The phase III study, to be initiated in 2023, will compare the efficacy of ganaplacide/lumefantrine-SDF to the current “gold standard” artemether-lumefantrine. The trial will be conducted in collaboration with the WANECAM 2 consortium and will include partner clinical sites in Burkina Faso, Mali, Gabon and Niger as well as other sites in sub-Saharan Africa.
Both phase II and phase III studies will receive funding from the European and Developing Countries Clinical Trials Partnership, which is supported by the European Union.
The decision to advance the candidate into phase III study follows the success of the phase II study, which was conducted in 524 adults and children with acute uncomplicated malaria due to Plasmodium falciparum infection. The ganaplacide/lumefantrine-SDF combination met the primary objective in both adults and children. In patients who received a once-daily dose of ganaplacide/lumefantrine-SDF during three days, the response to treatment was similar to the rate observed in patients who received twice-daily artemether-lumefantrine control therapy during three days.
The FDA granted Fast Track Designation and Orphan Drug Designation to the combination of ganaplacide and lumefantrine-SDF for the treatment of acute, uncomplicated malaria in August 2022.
Shares of Novartis have lost 0.2% so far this year against the industry’s rise of 8%.
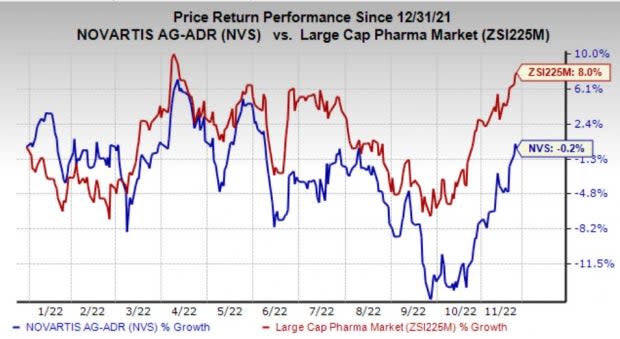
Image Source: Zacks Investment Research
Novartis’ performance in the third quarter was pretty ho-hum as earnings beat by a penny but revenues lagged due to generic competition. Novartis intends to transform into a pure-play Innovative Medicines company.
Novartis had stated that it would focus on further strengthening its position in five core therapeutic areas (Hematology, Solid Tumors, Immunology, Neuroscience and Cardiovascular).
The company earlier announced plans to spin off Sandoz into a new publicly traded standalone company following a strategic review. Due to industry-wide price competition among generic pharmaceutical companies and the consolidation of buyers, Sandoz experienced significant declines in sales and profits, particularly in the United States. The spin-off is likely to be completed by the first half of 2023.
The separation will enable Novartis to focus better on its legacy pharmaceutical business. Novartis had earlier spun off its eye care division, Alcon, into a new company.
Per a Bloomberg report, Novartis is also considering selling its ophthalmology and respiratory franchises.
Novartis’ ophthalmology franchise is primarily dependent on Lucentis, which the company markets with Roche RHHBY. However, Lucentis’ sales are declining in Europe and Japan due to biosimilar launches. The uptake of recently-approved Beovu, another ophthalmology drug, has not been encouraging due to concerns over potential side effects.
Per the report, the ophthalmology business alone could fetch about $5 billion.
Novartis’ respiratory franchise is primarily dependent on Xolair, which is also facing stiff competition. Novartis has a collaboration with Roche for Xolair.
Novartis currently carries a Zacks Rank #3 (Hold). A couple of better-ranked stocks in the healthcare space include Merck MRK and Biogen BIIB. Both the stocks carry a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Over the past 30 days, earnings estimates for Merck have increased by 7 cents to $7.38. Merck also surpassed estimates in all the trailing four quarters, with the average being 16.07%.
Over the past 30 days, earnings estimates for Biogen have increased by 30 cents to $16.98. Biogen also surpassed estimates in three of the trailing four quarters, with the average being 7.02%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Novartis AG (NVS) : Free Stock Analysis Report
Biogen Inc. (BIIB) : Free Stock Analysis Report
Roche Holding AG (RHHBY) : Free Stock Analysis Report
Merck & Co., Inc. (MRK) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 