Ocugen's (OCGN) COVID-19 Booster Effective Against Omicron
Ocugen, Inc. OCGN announced that its India-based partner, Bharat Biotech, reported positive data from the live virus neutralization assay evaluating a booster dose of its COVID-19 vaccine, Covaxin (BBV152). Data showed that administration of the booster dose following six months of the initial two-dose regimen of the vaccine led to robust neutralizing antibody responses against the two popular COVID-19 variants of concern — Omicron and Delta.
The live virus neutralization assay evaluated sera from individuals who received a booster of Covaxin against the two variants. The neutralizing activity of sera from individuals who received the booster was comparable to neutralizing activity of sera from individuals who received a booster of an mRNA-based COVID-19 vaccine namely — Pfizer PFE and BioNTech’s BNTX Comirnaty, and Moderna’s MRNA mRNA-1273.
The company stated that the neutralization antibodies were present in more than 90% of the individuals receiving the booster dose.
Last week, Bharat Biotech had posted promising results from a phase II study evaluating the booster dose of Covaxin in individuals aged 12 to 64 years on the pre-print server, medRxiv. Data from the study demonstrated a significant increase in neutralizing titers, compared to baseline (at six months following initial two-dose regimen), against key COVID-19 variants — Alpha, Beta, Delta and Delta plus.
Please note that Covaxin was developed by Bharat Biotech and is currently authorized for emergency use in 17 countries as well as by the World Health Organization. However, the vaccine is yet to receive approval/authorization in the United States. Meanwhile, more than 180 million doses of Covaxin have been administered outside of the United States.
Shares of Ocugen have gained 66.8% in the past year against the industry’s decrease of 29.8%.
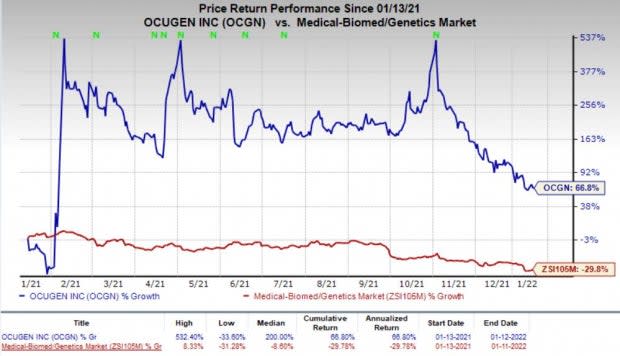
Image Source: Zacks Investment Research
Last month, Bharat Biotech posted positive results from a study of Covaxin in the age group of two to 18 years for COVID-19. The participants demonstrated a robust neutralizing antibody response and favorable safety profile when given a two-dose regimen administered 28-days apart. The children in this age group showed antibody responses comparable to adult data from a previous phase III study wherein greater than 93% reduction in severe disease was observed.
Although Ocugen and Bharat Biotech are progressing well with the development of their COVID-19 vaccine and its booster dose, they lag the leading COVID-19 vaccine makers.
Pfizer and BioNTech lead the race for the successful development of a COVID-19 vaccine, which is approved as a two-dose series to prevent COVID-19 in individuals 16 years of age and older in the United States. The booster dose of Pfizer and BioNTech’s COVID-19 vaccine has also been authorized for use in adults in the United States.
Moderna comes second with its mRNA-1273, which is also authorized for use in adults and adolescents. Moderna’s COVID-19 booster is also authorized for use in adults.
The combined sales of Pfizer, BioNTech and Moderna’s COVID-19 vaccines constituted a major chunk of total COVID-19 vaccine sales in 2021 and these vaccines are likely to maintain their lead in 2022 as well.
Ocugen, Inc. Price
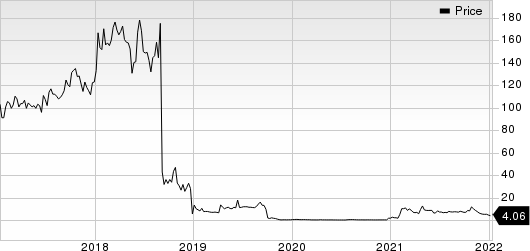
Ocugen, Inc. price | Ocugen, Inc. Quote
Zacks Rank
Ocugen currently carries a Zacks Rank #4 (Sell).
You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Pfizer Inc. (PFE) : Free Stock Analysis Report
Moderna, Inc. (MRNA) : Free Stock Analysis Report
Ocugen, Inc. (OCGN) : Free Stock Analysis Report
BioNTech SE Sponsored ADR (BNTX) : Free Stock Analysis Report
To read this article on Zacks.com click here.

 Yahoo Finance
Yahoo Finance 