AngioDynamics' (ANGO) AlphaVac System Gets CE Mark Approval
AngioDynamics, Inc. ANGO recently announced the European CE Mark approval for its AlphaVac F1885 System for the non-surgical removal of thrombi or emboli from the pulmonary arteries and the treatment of pulmonary embolism (PE).
The application of the AlphaVac F1885 System in the non-surgical excision of thrombi or emboli from the venous vasculature is enhanced by the CE Mark approval, which is likely a significant step forward in improving patient care and safety for endovascular therapies in the EU, a market with a higher prevalence of PE compared with the United States.
Price Performance
In the past six months, ANGO’s shares have lost 14.5% against the industry’s rise of 20.5%. The S&P 500 has gained 16.7% in the same time frame.
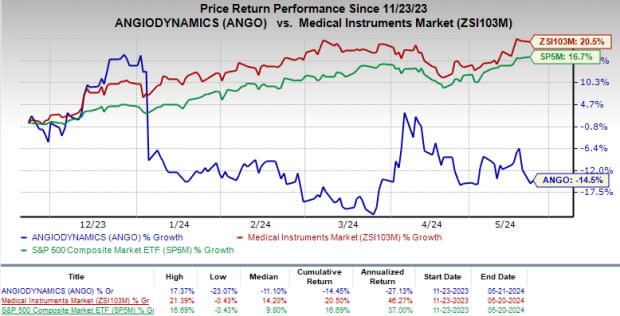
Image Source: Zacks Investment Research
More on AlphaVac F1885 System
At present, the AlphaVac F1885 System is authorized for utilization as an emergency first-line tool for managing PE and eliminating thromboemboli from the venous system. The components of the system include an ergonomic handle, an obturator, a waste bag assembly, and an 85-degree-angled 18F cannula. The APEX-AV study was created to provide safety and efficacy data for a clearance specific to PE.
More on the APEX-AV Study
In December 2023, AngioDynamics announced that patient enrollment in the single-arm Investigational Device Exemption study, Acute Pulmonary Embolism Extraction Trial with the AlphaVac System (APEX-AV), was completed. The study enrolled 122 patients with confirmed acute, intermediate-risk PE across 25 hospital-based sites in the United States to evaluate the AlphaVac F1885 System for the treatment of PE.
APEX-AV study's primary efficacy endpoint was the RV/LV ratio decrease from baseline to 48 hours after surgery. The rate of major adverse events within the first 48 hours, which included occurrences like severe bleeding and serious device-related clinical deterioration, pulmonary vascular injury, and cardiac injury, was the main safety goal. After the index procedure, patients were monitored for 30 days.
The results of the APEX-AV research demonstrated a quick clearing of the clot burden in the pulmonary arteries and a notable improvement in RV function.
The AlphaVac F1885 System's CE Mark increases treatment choices for medical providers in the EU by providing a tool that helps patients with PE have better right ventricular function and less thrombus burden.
Industry Prospects
Per a report by Data Bridge Market Research, the global pulmonary embolism market size, which was valued at $1.65 billion in 2022, is expected to reach more than $3.12 billion by 2030 at a growth rate of 8.3%.
The global pulmonary embolism market is driven by an increase in the prevalence of cancer. Another significant factor influencing the growth rate of the pulmonary embolism market is the rising healthcare expenditure.
Notable Developments
In April, AngioDynamics announced the FDA 510(k) clearance for its AlphaVac F1885 System for the treatment of pulmonary embolism.
AngioDynamics also announced that it has entered into a settlement agreement with Becton, Dickinson and Company, popularly known as BD, to resolve all outstanding patent litigation with C.R. Bard, Inc., an affiliate of BD.
AngioDynamics is likely to benefit from the settlement of this prolonged legal dispute to a successful resolution as it will lower the company’s ongoing legal expenses and terminate more than 10-year distraction.
In February, the company announced the completion of the sale of its PICC and Midline product portfolios to Spectrum Vascular for $45 million in cash.
AngioDynamics, Inc. Price
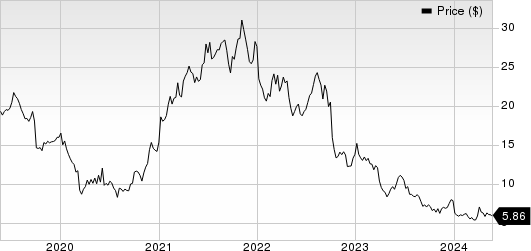
AngioDynamics, Inc. price | AngioDynamics, Inc. Quote
Zacks Rank & Stocks to Consider
ANGO carries a Zacks Rank #4 (Sell) at present.
Some better-ranked stocks in the broader medical space that have announced quarterly results are Align Technology, Inc. ALGN, Ecolab ECL and Boston Scientific Corporation BSX.
Align Technology, carrying a Zacks Rank of 2 (Buy), reported first-quarter 2024 adjusted earnings per share (EPS) of $2.14, beating the Zacks Consensus Estimate by 8.1%. Revenues of $997.4 million outpaced the consensus mark by 2.6%. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Align Technology has a long-term estimated growth rate of 6.9%. ALGN’s earnings surpassed estimates in three of the trailing four quarters and missed once, the average surprise being 5.9%.
Ecolab, carrying a Zacks Rank of 2 at present, has an estimated long-term growth rate of 13.3%. ECL’s earnings surpassed estimates in each of the trailing four quarters, with the average surprise being 1.7%.
Ecolab’s shares have rallied 33.8% against the industry’s 9.3% decline in the past year.
Boston Scientific reported first-quarter 2024 adjusted EPS of 56 cents, beating the Zacks Consensus Estimate by 9.8%. Revenues of $3.86 billion surpassed the Zacks Consensus Estimate by 4.9%. It currently carries a Zacks Rank #2.
Boston Scientific has a long-term estimated growth rate of 12.5%. BSX’s earnings surpassed estimates in all the trailing four quarters, the average surprise being 7.5%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Boston Scientific Corporation (BSX) : Free Stock Analysis Report
Ecolab Inc. (ECL) : Free Stock Analysis Report
AngioDynamics, Inc. (ANGO) : Free Stock Analysis Report
Align Technology, Inc. (ALGN) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 