Bristol Myers (BMY) Gains 24.2% YTD: What's in Store for 2023?
Bristol Myers BMY has put up a strong performance in 2022 as its shares have gained 24.2% so far against the industry’s decline of 19.7%.
In fact, like another biotech company Gilead Sciences, Inc. GILD, Bristol Myers is also among the handful of companies that have outperformed the industry and the S&P 500 Index in 2022.
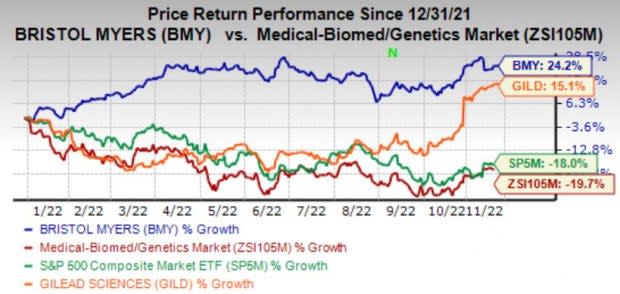
Image Source: Zacks Investment Research
Bristol-Myers beat third-quarter estimates for earnings and revenues. Sales however declined year over year due to the generic competition for Revlimid. Revlimid was the largest contributor to the company’s revenues, and the entry of generics for the company had adversely impacted sales.
Nevertheless, Bristol Myers seems to have more arrows in its quiver. As expected, the performance of blood thinner drug Eliquis and immuno-oncology drug Opdivo, along with encouraging pipeline progress, has maintained momentum.
Eliquis has delivered a stellar performance so far in 2022, propelled by increases in share in the novel oral anticoagulant market.
The label expansion of the Opdivo boosted sales, driven by higher demand across multiple indications, including the Opdivo+Yervoy-based combinations for non-small cell lung cancer (NSCLC), the Opdivo+Cabometyx combination for kidney cancer and the Opdivo-based therapies for various gastric and esophageal cancers.
The recent string of new drug approvals paved the way for an incremental stream of revenues. The FDA recently approved its experimental candidate deucravacitinib to treat adults with moderate-to-severe plaque psoriasis who are candidates for systemic therapy or phototherapy, under the brand name Sotyktu.
The FDA had earlier approved a new, first-in-class, fixed-dose combination of PD-1 inhibitor Opdivo and relatlimab (novel LAG-3-blocking antibody).
The uptake of the new product portfolio (primarily cell therapy products and Reblozyl) has also boosted sales.
However, per an article on Financial Times, Bristol Myers has now stated that it will cancel some drug development programs due to the U.S. government drug pricing rules that are due to be phased in from next year.
Per this article, BMY is reviewing its portfolio and expected that some drug candidates would not be funded as a result of reforms in the administration’s Inflation Reduction Act. This Act allows Medicare to negotiate for prescription drug prices and extend the expanded Affordable Care Act program for three years, through 2025.
Per BMY, oncology drugs/programs will mostly bear the brunt of the same. It remains to be seen how the company reshapes its pipeline development.
The diversification of the portfolio should enable the company to combat these challenges.
Earlier, per reports, pharma giant Eli Lilly LLY also stated that it is revaluating its pipeline development program and terminating development of a blood cancer drug owing to pricing reforms.
Lilly boasts a solid portfolio of core drugs in diabetes, autoimmune diseases and cancer.
Gilead’s shares gained 15.1% in the year so far as flagship HIV therapy Biktarvy maintains momentum for the company amid stiff competition.
Bristol Myers currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Bristol Myers Squibb Company (BMY) : Free Stock Analysis Report
Eli Lilly and Company (LLY) : Free Stock Analysis Report
Gilead Sciences, Inc. (GILD) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 