Novartis (NVS) Gets FDA Nod for Rare Blood Disorder Drug
Novartis NVS announced that the FDA approved iptacopan as the first oral monotherapy for the treatment of adults with paroxysmal nocturnal hemoglobinuria (“PNH”) under the brand name Fabhalta.
PNH is a rare, chronic and serious complement-mediated blood disorder. There is a significant need for treatments for this disease, as a large proportion of patients can remain anemic and dependent on blood transfusions despite anti-C5 treatments.
The FDA approval was based on the phase III APPLY-PNH trial, which evaluated the efficacy and safety of twice-daily, oral Fabhalta monotherapy (200 mg) for the treatment of PNH by assessing if switching to Fabhalta was superior to continuing anti-C5 therapies (Soliris and Ultomiris) in adult patients with residual anemia (Hb <10 g/dL) despite a stable regimen of anti-C5 treatment in the last six months prior to randomization.
Results of this study demonstrated that patients who switched to Fabhalta experienced superior increases of hemoglobin levels in the absence of red blood cell transfusions as compared with patients who continued with anti-C5 treatment. The approval was also supported by the APPOINT-PNH study in complement inhibitor-naïve patients.
Fabhalta has previously received Breakthrough Therapy Designation by the FDA in PNH.
The drug is also being evaluated for a range of complement-mediated diseases, including immunoglobulin A nephropathy (IgA nephropathy), C3 glomerulopathy (C3G), immune complex membranoproliferative glomerulonephritis and atypical hemolytic uremic syndrome.
The FDA also granted Breakthrough Therapy Designation to Fabhalta for C3G. Fabhalta also enjoys orphan drug designations from the FDA and European Medicines Agency in PNH and C3G. The European Medicines Agency has also granted orphan drug designation to Fabhalta for in IgAN.
Shares of Novartis have risen 6.9% year to date compared with the industry’s 5.4% growth.
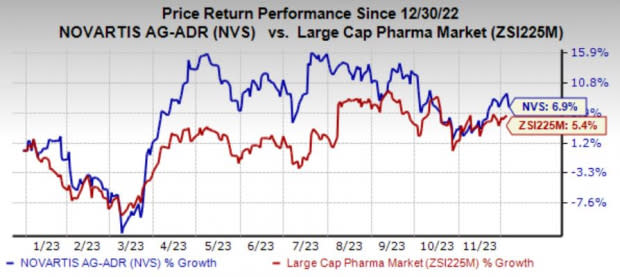
Image Source: Zacks Investment Research
Novartis is now a pure-play innovative medicine company following the spin-off of its generic division, Sandoz.
The approval of new drugs will expand its diverse pharmaceutical portfolio.
Earlier in 2023, Novartis acquired Chinook Therapeutics for $3.5 billion to strengthen its renal pipeline. The acquisition added two late-stage candidates, atrasentan and zigakibart, for the treatment of IgA nephropathy to NVS’ pipeline.
Strong performance of key drugs, a deep pipeline and a streamlined focus should help NVS maintain momentum amid competition.
We remind investors that Ultomiris, the first and only long-acting C5 complement inhibitor, was added to AstraZeneca’s AZN portfolio with the acquisition of erstwhile Alexion. It is approved in the U.S., EU and Japan for certain adults and children with PNH. Ultomiris is also approved by the FDA for the treatment of adult patients with generalized myasthenia gravis (gMG).
Strong sales of the drug have boosted AstraZeneca’s top line. The drug recorded sales of $2.1 billion in the first nine months of 2023, up 58% at the constant exchange rate. Ultomiris sales are benefiting from label expansion for the gMG indication, expansion into new markets and continued conversion from Alexion’s older drug, Soliris.
Zacks Rank and Stocks to Consider
Novartis currently has a Zacks Rank #3 (Hold).
A couple of better-ranked stocks in the overall healthcare sector are Entrada Therapeutics TRDA and Dynavax Technologies DVAX. While TRDA sports a Zacks Rank #1 (Strong Buy), DVAX carries a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.
Entrada’s loss per share estimate for 2023 has narrowed from $2.07 to 9 cents in the past 30 days. The same for 2024 has narrowed from $2.35 to $2.04 during the same time frame.
Dynavax’s loss per share estimate for 2023 has narrowed from 23 cents to 12 cents in the past 30 days. Earnings estimate for 2024 rose from 12 cents to 18 cents during the same period. Shares of DVAX have risen 29.3% year to date.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
AstraZeneca PLC (AZN) : Free Stock Analysis Report
Novartis AG (NVS) : Free Stock Analysis Report
Dynavax Technologies Corporation (DVAX) : Free Stock Analysis Report
Entrada Therapeutics, Inc. (TRDA) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 