Pharma Stock Roundup: AZN's 2030 Sales Target, PFE's New Cost Cut Plan & More
This week, AstraZeneca AZN announced its ambition to deliver $80 billion in revenues by 2030, which is significantly higher than $43.8 million generated in 2023. The FDA granted breakthrough therapy designation (“BTD”) to Roche’s RHHBY inavolisib combination regimen for a breast cancer indication. Eli Lilly LLY announced promising detailed data from a pivotal Crohn’s disease study on Omvoh (mirikizumab). Pfizer PFE announced a multi-year, multi-phase cost-reduction program targeting $1.5 billion in cost savings by 2027 in the first phase.
Recap of the Week’s Most Important Stories
AstraZeneca’s New Long-Term Revenue Target: AstraZeneca announced its long-term plan to generate $80 billion in total revenues by 2030. AstraZeneca plans to launch 20 new medicines by 2023, many of which will have the potential to generate more than $5 billion in peak-year revenues. AstraZeneca will continue investing in disruptive innovation and transformative new technologies and platforms to discover novel medicines.
AstraZeneca said its oncology, biopharmaceuticals and rare disease drugs will be the key drivers of top-line growth. AstraZeneca also said it expects to generate a mid-30s percentage core operating margin by 2026 and beyond. In 2023, the core operating margin was 32%.
Lilly’s Detailed Data From Omvoh Study in Crohn’s Disease: Lilly announced additional data from the pivotal phase III VIVID 1 study evaluating Omvoh (mirikizumab) for treating moderately to severely active Crohn's disease. Lilly has already filed regulatory applications in the United States and Europe seeking approval for Omvoh for its second inflammatory bowel disease (IBD) indication, Crohn's disease, earlier this year based on data from the VIVID 1 study. Omvoh was approved for its first IBD indication, ulcerative colitis, in the United States, Europe and Japan in 2023.
Lilly previously announced that the VIVID-1 study met both co-primary endpoints (clinical remission and endoscopic response) and all major secondary endpoints at week 52 compared to placebo. In the latest release, Lilly said that similar response rates and treatment effects were observed in patients with no prior biologic failure (bio-naïve) as well as those with previous biologic failure, the latter being a difficult-to-treat population.
At one year, 54.1% and 48.4% of patients treated with Omvoh achieved clinical remission and endoscopic response, respectively. Importantly, the majority of patients who achieved either of these endpoints achieved both together. Among the Omvoh-treated patients, 56.7% of bio-naïve and 51.2% of bio-failed patients achieved clinical remission at week 52.
Pfizer Multi-Year Cost Savings Plan: Pfizer, in a securities filing, announced a multi-year cost reduction program, which will include operational efficiencies, network structure changes and product portfolio enhancements. Under the first phase of this multi-phased effort, Pfizer expects to achieve approximately $1.5 billion of cost savings by the end of 2027 through operational efficiencies. Some of these cost savings are expected to be realized in 2025. The one-time costs related to the first phase, which will include severance and implementation costs, will be $1.7 billion. This will be recorded primarily in 2024. Pfizer, however, did not mention the number of jobs it expects to cut. In 2023, Pfizer announced a $4 billion cost-cutting plan.
FDA’s Breakthrough Tag to Roche’s Breast Cancer Candidate: The FDA granted BTD to Roche’s oral candidate inavolisibplus Ibrance and fulvestrant for advanced hormone receptor-positive, HER2-negative breast cancer with a PIK3CA mutation. The designation is based on data from the phase III INAVO120 study. The study showed that the inavolisib combination regimen reduced the risk of disease worsening or death (progression-free survival) by 57% compared to Ibrance plus fulvestrant.
The NYSE ARCA Pharmaceutical Index rose 0.4% in the last five trading sessions.
Large Cap Pharmaceuticals Industry 5YR % Return
Large Cap Pharmaceuticals Industry 5YR % Return
Here’s how the eight major stocks performed in the last five trading sessions.
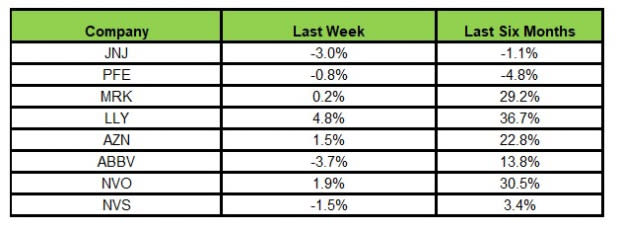
Image Source: Zacks Investment Research
In the last five trading sessions, Lilly rose the most (4.8%), while AbbVie declined the most (3.7%).
In the past six months, Lilly has risen the most (36.7%), while Pfizer has declined the most (4.8%).
(See the last pharma stock roundup here: BAYRY’s Q1 Earnings, JNJ’s New Buyout, Pipeline Updates)
What's Next in the Pharma World?
Watch this space for regular pipeline and regulatory updates next week.
Pfizer, Lilly and AstraZeneca have a Zacks Rank #3 (Hold) each, while Roche has a Zacks Rank of 4 (Sell).
You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
AstraZeneca PLC (AZN) : Free Stock Analysis Report
Roche Holding AG (RHHBY) : Free Stock Analysis Report
Pfizer Inc. (PFE) : Free Stock Analysis Report
Eli Lilly and Company (LLY) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 