Sage (SAGE) Down 20% on Parkinson's Study Missing Primary Goal
Shares of Sage Pharmaceuticals SAGE dropped 19.6% on Wednesday after management shared topline results from the phase II PRECEDENT study (n=86) evaluating its lead neuropsychiatric candidate dalzanemdor in patients with mild cognitive impairment (MCI) associated with Parkinson’s disease (PD).
The study failed to achieve its primary endpoint of demonstrating a statistically significant difference from baseline in participants treated with orally-administered dalzanemdor, when compared to placebo. Per management, the analyses of data also did not indicate any meaningful differences versus placebo in the other exploratory endpoints, such as SCOPA-Cog.
Management also pointed out that 48 patients, which represent more than half of the total study participants, experienced treatment emergent adverse events (TEAEs) ranging from mild to moderate in severity.
Based on these results, management has decided not to pursue the development of dalzanemdor in PD indication. The share price was down likely due to this reason.
Year to date, shares of Sage have lost 42.0% compared with the industry’s 11.2% fall.
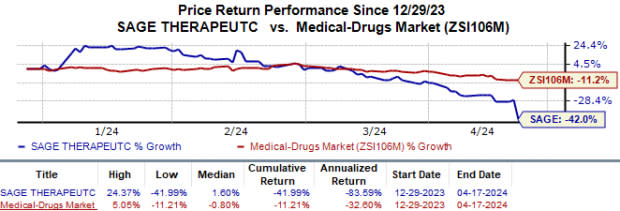
Image Source: Zacks Investment Research
Apart from PD, Sage Therapeutics is evaluating dalzanemdor as a potential treatment for patients with cognition dysfunction caused by Huntington’s disease (HD) across three ongoing clinical studies — two phase II studies (DIMENSION and SURVEYOR) and a phase III study (PURVIEW). The candidate is also being evaluated in a mid-stage study for treating cognitive issues associated with Alzheimer’s disease. Data from these studies are expected throughout this year.
SAGE has been under pressure since last year when the FDA decided on zuranolone in postpartum depression (PPD) and major depressive disorder (MDD) indications. While the agency approved the drug for treating PPD, it issued a complete response letter (CRL) in the MDD indication. The drug will be marketed in PPD indication under the trade name Zurzuvae.
Per the CRL, the FDA stated that the NDA filing did not provide substantial evidence of effectiveness to support the approval of zuranolone. Hence, an additional study or studies will be needed.
Though the approval in PPD was positive, MDD is a much larger and more lucrative market opportunity. In addition, the FDA approval for PPD also comes with a back box warning – advising patients not to drive or operate heavy machinery for at least 12 hours after taking Zurzuvae.
Sage Therapeutics has developed Zurzuvae in collaboration with Biogen BIIB. Currently, Sage and Biogen are evaluating the further course of action. Management is yet to notify a future course of action in this regard.
Following the above setbacks and to support Zurzuvae’s commercial launch, Sage Therapeutics implemented a strategic reorganization plan. Based on this plan, the company reduced its workforce by around 40%, focused on the pipeline development of dalzanemdor and lead neurology program, SAGE-324. Like Zurzuvae, SAGE-324 is also being developed in collaboration with Biogen.
Sage Therapeutics, Inc. Price
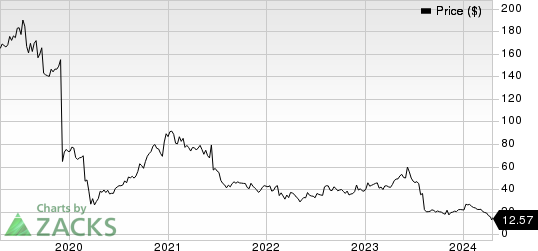
Sage Therapeutics, Inc. price | Sage Therapeutics, Inc. Quote
Zacks Rank & Key Picks
Sage currently carries a Zacks Rank #3 (Hold). A couple of better-ranked stocks in the overall healthcare sector include ADMA Biologics ADMA and ANI Pharmaceuticals ANIP, each sporting a Zacks Rank #1 (Strong Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.
In the past 60 days, estimates for ADMA Biologics’ 2024 earnings per share (EPS) have risen from 22 cents to 30 cents. During the same period, EPS estimates for 2025 have improved from 32 cents to 50 cents. Year to date, shares of ADMA have surged 33.6%.
Earnings of ADMA Biologics beat estimates in three of the last four quarters while meeting the same on one occasion. ADMA delivered a four-quarter average earnings surprise of 85.00%.
In the past 60 days, estimates for ANI Pharmaceuticals’ 2024 EPS have risen from $4.12 to $4.43. Meanwhile, during the same period, EPS estimates for 2025 have improved from $4.80 to $5.04. Year to date, shares of ANIP have risen 18.8%.
Earnings of ANI Pharmaceuticals beat estimates in each of the last four quarters. ANI delivered a four-quarter average earnings surprise of 109.06%.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Biogen Inc. (BIIB) : Free Stock Analysis Report
ANI Pharmaceuticals, Inc. (ANIP) : Free Stock Analysis Report
ADMA Biologics Inc (ADMA) : Free Stock Analysis Report
Sage Therapeutics, Inc. (SAGE) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 