Trethera Receives U.S. Patent Office Notice of Allowance Covering TRE-515 Structural Claims, Extending Protections to Late 2041

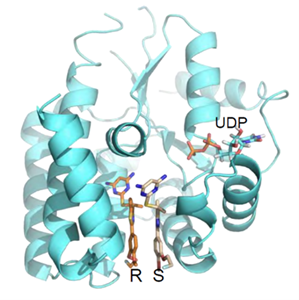
Figure 1
LOS ANGELES, Aug. 17, 2022 (GLOBE NEWSWIRE) -- Trethera Corporation (“Trethera”), a clinical stage biopharmaceutical company committed to developing novel drugs targeting nucleotide metabolism for the treatment of cancer and autoimmune diseases, announces today that the United States Patent and Trademark Office (USPTO) issued a Notice of Allowance for a composition of matter patent covering the polymorphic form of TRE-515. The resulting US patent will extend the patent protection for TRE-515 in the United States by seven years through November 2041. TRE-515 is a first-in-class drug targeting the enzyme deoxycytidine kinase (dCK) and currently in Phase 1 clinical trials.
“We are extremely pleased with this expansion of the TRE-515 patent portfolio,” said Dr. Ken Schultz, Trethera CEO and patent co-inventor. “Trethera’s ownership of this patent is another major step in the commercialization of TRE-515, providing 19 years of composition of matter patent life when our final dose escalation patient enrolls this year. The patent claims cover the unique TRE-515 chemical structure, whether used in the treatment of cancer, autoimmune, or other diseases.”
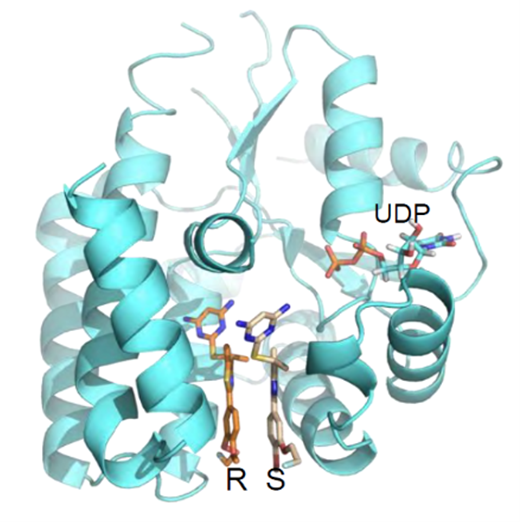
Figure 1. Co-crystal 3D structure of the drug bound to the target enzyme, dCK, at the deoxycytidine binding site.
Trethera is currently enrolling patients in a Phase 1 dose escalation trial to evaluate TRE-515 monotherapy in patients with various solid tumors. Once a day oral doses have escalated from an initial 40mg dose to the current 240mg dose level. Trethera reported in June 2022 that TRE-515 was well tolerated by all enrolled patients (N=10) with adverse events being transient and manageable. Furthermore, 50% of the patients who completed their second staging CT scan (N=6) before June showed stable disease. The dose escalation portion of the trial is expected to enroll the final patient before year end.
Trethera’s outside intellectual property counsel, Wilson Sonsini Goodrich & Rosati, led the patent prosecution. This Notice of Allowance concludes the substantive examination period of the patent application and will result in the issuance of a patent after USPTO administrative fees and processes are completed in the coming months. Similar patent application efforts are underway globally, including the major pharmaceutical markets of Europe and Japan. Trethera’s TRE-515 patent portfolio includes other granted US and global patents and pending applications directed to specialized methods of use for cancer and autoimmune diseases.
About Trethera
Trethera is a clinical stage privately held biopharmaceutical company dedicated to pioneering the development of novel treatments for autoimmune diseases and cancers. Founded by prominent UCLA scientists, Trethera is led by experienced management and board members. Trethera's innovative approach to targeting nucleotide metabolism led to the development of TRE-515, an orally taken capsule twice designated by the FDA as an Orphan Drug. TRE-515 is a first-in-class clinical stage drug that inhibits deoxycytidine kinase (dCK), the rate-limiting enzyme in the nucleoside salvage pathway, one of two biosynthetic pathways that generate DNA precursors. It is believed that some forms of cancer may be preferentially dependent on the salvage pathway to support tumor growth, and certain autoimmune diseases, such as multiple sclerosis, might also respond to TRE-515 treatment. Trethera is developing TRE-515 for use as a monotherapy or in combination, to precisely target a metabolic vulnerability of cancer or autoimmune diseases that will transform outcomes for patients.
For more information, please visit us at trethera.com or e-mail Investor Relations at ir@trethera.com.
Note on Forward-Looking Statements
All statements other than statements of historical facts included in this press release that address activities, events or developments that Trethera believes or anticipates will or may occur in the future are “forward-looking statements,” which may often, but not always, be identified by the use of such words as "may," "might," "will," "will likely result," "would," "should," "estimate," "plan," "project," "forecast," "intend," "expect," "anticipate," "believe," "seek," "continue," "target" or the negative of such terms or other similar expressions. Although Trethera has a reasonable basis for the forward-looking statements contained herein, Trethera cautions that such statements are based on current expectations about future events and are subject to risks, uncertainties and factors relating to medical and scientific research, all of which are difficult to predict and many of which are beyond Trethera’s control, that may cause actual results to differ materially from those expressed or implied by the forward-looking statements in this press release. These potential risks and uncertainties include, without limitation: the extent to which development of any novel cancer therapies or therapies for autoimmune diseases succeeds; whether Trethera would obtain the necessary regulatory approvals to commence human trials or commercialize TRE-515 or any novel therapies resulting from such research; Trethera successfully implementing its growth strategy, including that relating to its disease therapies; the effects of the global Covid-19 pandemic; changes in economic conditions; competition; and risks and uncertainties applicable to the business of Trethera. The statements in this press release speak only as of the date hereof and Trethera does not undertake any obligation to update, amend or clarify these forward-looking statements whether as a result of new information, future events or otherwise. The Company intends that all forward-looking statements be subject to the safe-harbor provisions of the Private Securities Litigation Reform Act of 1995.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/bb4121f3-9cd3-4e04-bea7-06c8590ea447


 Yahoo Finance
Yahoo Finance 