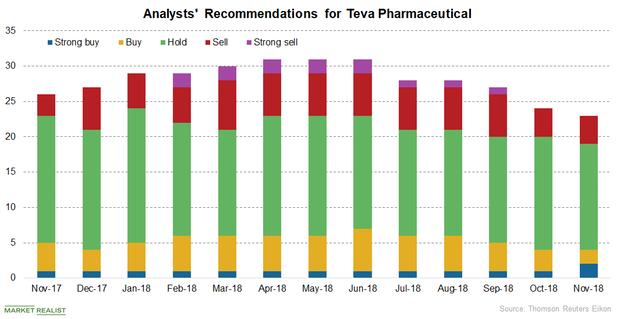
How Wall Street Analysts View Teva Pharmaceutical
In October, the FDA’s Oncologic Drugs Advisory Committee unanimously voted to recommend approving Teva Pharmaceutical’s (TEVA) and Celltrion’s Biologics License Application for CT-P10, a proposed biosimilar to Roche’s (RHHBY) Rituxan. The recommended approval of CT-P10 was for three indications: as a monotherapy for relapsed or refractory B-cell non-Hodgkin’s lymphoma as a first-line treatment for B-cell NHL in combination with chemotherapy as a second-line monotherapy for non-progressing B-cell NHL after cyclophosphamide-, vincristine-, or prednisone-based chemotherapy

 Yahoo Finance
Yahoo Finance 