Biophytis (BPTS) to Begin Phase II Obesity Study, Stock Up
Biophytis S.A.’s BPTS shares surged 63.1% in the after-market hours on Apr 8, after the company announced the launch of a phase II study called OBA, with its pipeline candidate, BIO101 (20-hydroxyecdysone), to treat obesity in combination with GLP-1 receptor agonists (GLP-1 RA).
Biophytis’ BIO101 is the first oral daily MAS receptor activator that is expected to prevent extensive muscle loss due to obesity treatment. Currently, approved GLP-1 RA drugs for obesity, indicated in conjunction with dieting, are very effective in inducing significant weight loss. However, the treatment regime can also lead to loss of muscle mass and function for which no therapies are available presently.
Per Biophytis, up to 40% of the total weight loss comes from muscle, which is a problem as the role of muscle tissues is central in controlling metabolism, on top of its motor function.
The obesity market has garnered much interest in 2023, ever since GLP-1 drugs like Novo Nordisk’s NVO Wegovy (semaglutide 2.4 mg) and Eli Lilly’s LLY Zepbound (tirzepatide) received FDA approval for chronic weight management. NVO’s Wegovy is a GLP-1 agonist while on the other hand, LLY’s Zepbound is a dual GIP and GLP-1 receptor agonist.
Biotech firms have been competing fiercely throughout 2023 to enter the obesity market due to the huge untapped market opportunity that it represents amid increasing demand. Innovative obesity treatments offer an increasingly lucrative market opportunity.
Biophytis referred to research conducted by Goldman Sachs stating that more than 15 million adults in the United States will be treated with an anti-obesity medication by 2030, which is expected to represent a massive market opportunity of approximately $100 billion.
Year to date, shares of BPTS have plunged 52.3% compared with the industry’s 5.7% decline.
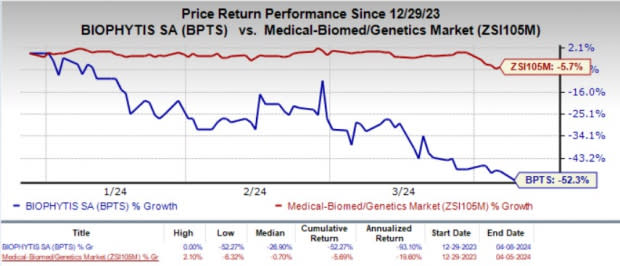
Image Source: Zacks Investment Research
The company has further reported that treatment with BIO101 has shown metabolic effects on muscle and fat tissues in preclinical obesity studies.
BIO101 is also currently being evaluated in a phase IISARA-INT study for obese sarcopenic patients. The therapeutic benefits of the candidate can also benefit this additional patient population, potentially leading to improved mobility and muscle strength.
Additionally, BPTS stated that the previously conducted Quinolia study in obese patients during hypocaloric dieting has already validated the promising effects of the 20-hydroxyecdysone molecule on muscle strength and fat mass loss.
The company expects to initiate the phase II OBA clinical study of BIO101 in mid-2024, subject to regulatory approvals, with the first patients expected to be treated in the second half of 2024. The intended patient population for the OBA study is obese patients treated with GLP-1 RAs and following hypocaloric dieting. Top-line efficacy results from the study are anticipated in 2025.
Biophytis SA Sponsored ADR Price and Consensus
Biophytis SA Sponsored ADR price-consensus-chart | Biophytis SA Sponsored ADR Quote
Wegovy is Novo Nordisk’s blockbuster chronic weight management injection approved for adults with obesity or overweight. The drug received FDA approval in 2021 and has been witnessing solid uptake ever since, driven by increasing demand worldwide. In 2023, NVO’s Wegovy generated sales worth DKK 31.3 billion, representing year-over-year growth of 407% on a reported basis and 420% at a constant exchange rate.
Eli Lilly initially received FDA approval for tirzepatide under the brand name Mounjaro in 2022 to treat adults with type II diabetes. In November 2023, LLY received FDA approval for the same tirzepatide formulation, under the brand name Zepbound, for chronic weight management in adults with obesity or overweight. Approval of Zepbound is expected to help LLY rake in billions of dollars.
Amgen AMGN is also currently developing its own GLP-1 RA drugs for obesity management. Amgen has two investigational candidates, maridebart cafraglutide and AMG 786, in a phase II study and a phase I study, respectively, for the obesity indication. Out of these two candidates, maridebart cafraglutide is particularly intriguing, having a unique mechanism of action. This investigational drug activates the GLP-1 receptor and inhibits the GIP receptor.
Zacks Rank
Biophytis currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Novo Nordisk A/S (NVO) : Free Stock Analysis Report
Eli Lilly and Company (LLY) : Free Stock Analysis Report
Amgen Inc. (AMGN) : Free Stock Analysis Report
Biophytis SA Sponsored ADR (BPTS) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 