Insulet (PODD) Reports Pivotal Extended Phase Data for Omnipod 5
Insulet Corporation PODD recently presented new Omnipod 5 Automated Insulin Delivery System (Omnipod 5) study outcomes for children with type 1 diabetes, aged 2 through 5.9 years, at the American Diabetes Association 82nd Scientific Sessions. The Omnipod 5 is the first tubeless, wearable automated insulin delivery (AID) system in the United States. It was previously shown to be safe and effective in the original three-month pivotal study on 80 very young children with type 1 diabetes.
The latest pivotal extension data for pre-school children demonstrated significantly improved time in range and lower HbA1c in children with type 1 diabetes in the above-mentioned age group throughout 12 months of Omnipod 5 usage. These results suggest that Omnipod 5 is an ideal option for AID in this vulnerable age group to alleviate the burden of diabetes for families and caregivers and optimize glycemic outcomes.
The company also plans to present additional clinical data revealing enhanced outcomes for adults and children with type 1 diabetes and adults with type 2 diabetes with the Omnipod 5 System.
Overview of Preschool Extension Trial Data
All participants from the original pivotal trial continued using the Omnipod 5 System for an ongoing 12+ month extension phase. The study results were examined after all participants had used Omnipod 5 for 12 months (three months in the pivotal trial and nine months in the extension phase). Throughout the 12 months of system use, participants continued to exhibit significantly improved outcomes compared with baseline, including lower HbA1c and more time in range with no occurrences of DKA or severe hypoglycemia.
Post twelve months of Omnipod 5 use, the average HbA1c was 6.9% compared with 7.4% at baseline and 6.9% at the end of the three-month pivotal trial. During 10-12 months of use, the percent time in range was found to be 67.6% versus 57.2% in standard therapy and 68.1% in the three-month pivotal trial. Further, the percent time in hypoglycemia (<70 mg/dL) was also reduced when compared to standard therapy.
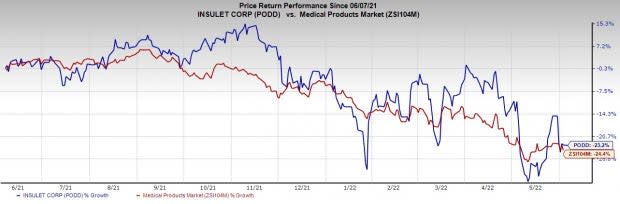
Image Source: Zacks Investment Research
Per management, the safety and improved glycemic outcomes from the initial three-month pivotal study in pre-school aged children were extended for an additional nine months. This is suggestive of the potential long-term benefit of the Omnipod 5 System in very young people with type 1 diabetes.
Industry Prospects
Per a report published in BUSINESS WIRE, the global Automated Insulin Delivery System market is expected to see a CAGR of 9.59% by 2026. Factors such as the increasing prevalence of diabetes, several awareness campaigns and growing investment in research and development for new technology and automation in medical devices can be attributed to market growth.
Given the market prospects, the latest extension phase data for pre-school children seem strategic and well-timed.
Other Notable Developments
In June 2022, Insulet noted its plans to construct its new manufacturing facility in Johor Bahru, Malaysia. The company expects the facility to be operational by the middle of 2024 and strengthen its global manufacturing capabilities. This new manufacturing facility will have approximately 400,000 square feet of space. The company aims to produce Omnipod Insulin Management System in the facility.
Added to this, Insulet also made several efforts to reinforce its global manufacturing scale and flexibility during the first quarter of 2022. In this regard, the company has optimized operations in China by consolidating its production into a newer lower-cost facility. These initiatives are anticipated to support Insulet’s international expansion strategy, ensure product supply and drive margin improvement over time.
Share Price Performance
The stock has outperformed its industry over the past year. It has declined 23.2% compared to the industry’s 24.4% fall.
Zacks Rank and Key Picks
Currently, Insulet carries a Zacks Rank #3 (Hold).
A few better-ranked stocks in the broader medical space are AMN Healthcare Services, Inc. AMN, Medpace Holdings, Inc. MEDP and UnitedHealth Group Incorporated UNH.
AMN Healthcare has a long-term earnings growth rate of 1.1%. The company surpassed earnings estimates in the trailing four quarters, delivering a surprise of 15.6%, on average. It currently flaunts a Zacks Rank #1 (Strong Buy). You can see the complete list of today’s Zacks #1 Rank stocks here.
AMN Healthcare has outperformed its industry in the past year. AMN has gained 4.8% against the industry’s 64.1% fall.
Medpace has a historical growth rate of 27.3%. Medpace’s earnings surpassed estimates in the trailing four quarters, the average surprise being 17.1%. It currently has a Zacks Rank #2 (Buy).
Medpace has outperformed its industry in the past year. MEDP has declined 14.2% compared with the industry’s 64.1% fall.
UnitedHealth has an estimated long-term growth rate of 14.8%. UnitedHealth’s earnings surpassed estimates in the trailing four quarters, the average surprise being 3.7%. It currently carries a Zacks Rank #2.
UnitedHealth has outperformed the industry over the past year. UNH has gained 21.9% compared with 19% industry growth in the said period.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
UnitedHealth Group Incorporated (UNH) : Free Stock Analysis Report
AMN Healthcare Services Inc (AMN) : Free Stock Analysis Report
Insulet Corporation (PODD) : Free Stock Analysis Report
Medpace Holdings, Inc. (MEDP) : Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research

 Yahoo Finance
Yahoo Finance 