Ionis (IONS) Q1 Earnings Surpass Estimates, Sales Match
Ionis Pharmaceuticals IONS reported a loss of 87 cents per share for first-quarter 2023, much narrower than the Zacks Consensus Estimate as well as our estimate of a loss of $1.00 per share. In the year-ago quarter, the company had reported a loss of 46 cents per share.
The bottom line includes compensation expense related to equity awards. Excluding these special items, the adjusted loss per share was 68 cents per share versus 27 cents per share in the year-ago quarter.
Total revenues were $131 million in the first quarter, which came in line with the Zacks Consensus Estimate as well as our estimate. Revenues declined 7.7% year over year.
Quarter in Detail
Ionis has licensed Spinraza to Biogen BIIB. Biogen is responsible for commercializing Spinraza, approved for treating spinal muscular atrophy, or SMA, worldwide. Ionis receives royalties from Biogen on Spinraza’s sales.
Commercial revenues were $68 million in the first quarter, down 5.6% year over year.
Commercial revenues from Spinraza royalties were $50 million, down 7.4% year over. Spinraza product sales (recorded by Biogen) were hurt by lower new patients starts in the United States, currency headwinds and unfavorable channel dynamics in the first quarter.
Revenues from Tegsedi and Waylivra from distribution fees were $7 million compared with $6 million in the year-ago quarter. License and royalty revenues were $11 million in the quarter compared with $12 million in the year-ago quarter.
R&D revenues of $63 million were less than the year-ago revenues of $70 million. The R&D revenues in the quarter included a payment from AstraZeneca AZN for costs shared for eplontersen development as well as some milestone payments from partners Biogen and GSK GSK.
Adjusted operating costs were up 26% year over year to $218 million in the first quarter, mainly due to higher R&D costs, as the company rapidly advanced its wholly-owned late-stage pipeline and increased go-to-market activities for eplontersen, olezarsen and donidalorsen.
2023 Guidance
Ionis reaffirmed its previously issued financial guidance for 2023. The company expects total revenues to be more than $575 million in 2023. Its adjusted operating loss is expected to be less than $425 million.
Adjusted operating expense is expected to be in the range of $970-$995 million. Adjusted operating costs are expected to increase slightly in the second quarter and then gradually increase in the second half. R&D costs are expected to increase in the range of 20-25% year over year in 2023. SG&A costs are expected to increase approximately $35 million year over year.
The company expects its cash and investment to be approximately $2 billion in 2023.
Ionis’ shares were down 2.2% on Wednesday in response to the earnings release. In the past year, Ionis’ shares have declined 8.8% compared with the industry’s decrease of 7.9%.
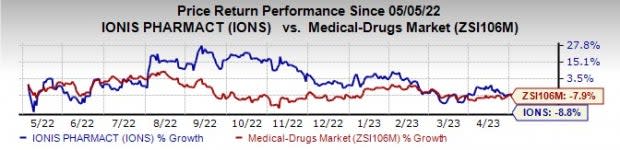
Image Source: Zacks Investment Research
Pipeline Update
Ionis has phase III studies ongoing for seven medicines (internal as well as partnered) across nine indications.
Some of these candidates are tofersen for SOD1-ALS (amyotrophic lateral sclerosis), pelacarsen for cardiovascular disease due to elevated Lp(a) levels, olezarsen for familial chylomicronemia syndrome (FCS) and severe hypertriglyceridemia, eplontersen for TTR amyloidosis, ION363 for ALS, with mutations in the fused in sarcoma gene, or FUS; donidalorsen for hereditary angioedema and bepirovirsen for chronic hepatitis B.
Biogen, AstraZeneca, Novartis and GSK are its partners for tofersen, eplontersen, pelacarsen and bepirovirsen, respectively. Ionis and AstraZeneca’s new drug application seeking approval of eplontersen for polyneuropathy caused by hereditary TTR amyloidosis (ATTRv-PN) is under review with the FDA with a decision expected on Dec. 22, 2023. In the phase III NEURO-TTRansform study of eplontersen in patients with ATTRv-PN, eplontersen demonstrated robust and sustained reductions in serum TTR and significant improvements in measures of neuropathy and quality of life with a substantial number of patients demonstrating improvement at both week 35 and week 66.
Ionis and AstraZeneca are also developing eplontersen for the treatment of cardiomyopathy in the phase III CARDIO-TTRansform study. Enrollment in the study is expected to be completed in mid-2023.
In April, the FDA granted accelerated approval to Biogen/Ionis’ tofersen for treating superoxide dismutase 1-amyotrophic lateral sclerosis. The drug will be marketed by the trade name of Qalsody. A regulatory application for tofersen is under review in the EU.
In the quarter, partner GSK advanced bepirovirsen into phase III development in patients with chronic hepatitis B.
Ionis is advancing and expanding its wholly-owned pipeline to drive future revenue growth. Many of these candidates — olezarsen, ION-363 and donidalorsen — are being evaluated in late-stage studies. Data from the phase III BALANCE study of olezarsen in FCS is expected in the second half of this year.
Zacks Rank & Stocks to Consider
Ionis currently has a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Ionis Pharmaceuticals, Inc. Price, Consensus and EPS Surprise
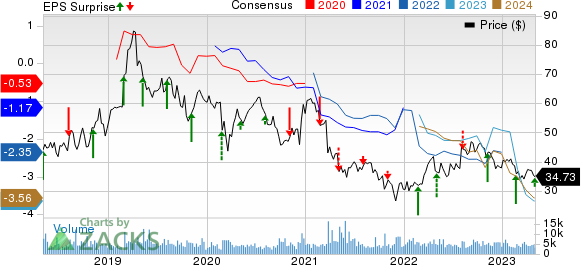
Ionis Pharmaceuticals, Inc. price-consensus-eps-surprise-chart | Ionis Pharmaceuticals, Inc. Quote
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
AstraZeneca PLC (AZN) : Free Stock Analysis Report
GSK PLC Sponsored ADR (GSK) : Free Stock Analysis Report
Biogen Inc. (BIIB) : Free Stock Analysis Report
Ionis Pharmaceuticals, Inc. (IONS) : Free Stock Analysis Report

 Yahoo Finance
Yahoo Finance 